COLUMBUS, Ohio. (Ivanhoe Newswire)—According to the FDA, as of late last year there were 370 active trials for therapies to treat COVID-19. For some critically ill patients, finding treatment has been trial and error. Therapies that worked for some made no impact on others. One Connecticut family says an internet search for options led them to an investigational drug, ZYESAMI, that they say saved their loved one’s life.
Baskets of get-well wishes for 63-year-old Michael DiDonato and his wife Lori. Both were hospitalized with COVID on the same day. Lori got better; Michael wound up on a ventilator … hovering near death.
“There were a couple of days like that, where we went to bed thinking, you know, preparing ourselves, we might get a call tonight, you know, we have to be prepared for that,” recalled Michael’s wife, Lori DiDonato.
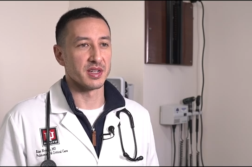
“It really was that critical,” stated Daniel A. Gerardi, MD, FCCP, Chief, Section of Pulmonary, Critical Care, and Sleep Medicine at Saint Francis Hospital in Hartford, CT.
Michael’s son began searching for survivor stories and read about a Florida doctor who recovered after treatment with an experimental drug known now as ZYESAMI.
“I looked into it, we weren’t gonna just jump on the bandwagon with some kind of quack thing,” Lori shared.
In order to get access to the therapy, the DiDonato’s physician had to ask the drug company to allow compassionate use.
“This was a novel therapy that had some good data, preliminary data, about its anti-inflammatory effect, and evasive dilator effect, so that it might improve a patient’s oxygenation,” explained Dr. Gerardi.
The company rushed ZYESAMI to St. Francis Hospital where doctors infused the drug for 12 hours, three days in a row. After a second round of the therapy, Michael began to respond.
“We couldn’t believe it. We were so joyful,” Lori exclaimed.
“Sounds like it was a Hail Mary,” Michael DiDonato shared.
Michael’s voice is weak and he uses a walker, but he’s thankful to be here … and working to get stronger every day.
The DiDonatos say their entire family played a huge role in coordinating the quick delivery of the drug to the hospital, and they says they’re convinced ZYESAMI played a role in Michael’s recovery. Dr. Gerardi says there’s still not enough science to confirm that the drug worked. A phase three clinical trial of ZYESAMI started at the end of April. Michael was not part of that trial.
Contributors to this news report include: Cyndy McGrath, Executive & Field Producer; Kirk Manson, Videographer; Roque Correa, Editor.
To receive a free weekly e-mail on Medical Breakthroughs from Ivanhoe, sign up at: http://www.ivanhoe.com/ftk
Sources:
https://www.fda.gov/media/136832/download
MEDICAL BREAKTHROUGHS
RESEARCH SUMMARY
TOPIC: ZYESAMI: INVESTIGATIONAL DRUG FOR COVID?
REPORT: MB #4912
BACKGROUND: A newly identified coronavirus, SARS-CoV-2, has caused a worldwide pandemic of respiratory illness, called COVID-19. Symptoms include cough, fever, shortness of breath, muscle aches, sore throat, unexplained loss of taste or smell, diarrhea and headache. COVID-19 can be severe, and some cases has caused death. COVID-19 is spread through droplets released into the air when an infected person coughs or sneezes. As of now, there is no specific treatment for the virus. People who have COVID-19 are most commonly treated with supportive measures that relieve symptoms and are often mechanically ventilated for extended periods of time. For severe cases, there are additional options for treatment, including research drugs and therapeutics. According to the Food and Drug Administration, there were 370 active trials for therapies to treat COVID-19 as of late 2020.
(Sources: https://www.fda.gov/media/136832/download, https://www.hopkinsmedicine.org/health/conditions-and-diseases/coronavirus)
ZYESAMI: Dosing of the first patient in a phase 3 clinical trial of ZYESAMI, or aviptadil acetate, which is a synthetic version of a vasoactive peptide, was announced by the National Institutes of Health in April 2021The trial, designated as ACTIV-3b: Therapeutics for Severely Ill Inpatients With COVID-19. The study will be carried out across the United States as part of the NIH Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV) public-private partnership to prioritize and accelerate development of the most promising COVID-19 treatments. Dr. Anthony Fauci elaborated on the need for new COVID-19 therapeutics and stated that the trial is randomized, blinded and placebo controlled.
NEW TECHNOLOGY: Daniel A. Gerardi, MD, FCCP, Chief of pulmonary, critical care, and sleep medicine at St. Francis Hospital, in Hartford, Connecticut treated one patient with ZYESAMI who was able to receive compassionate use for the drug to treat COVID prior to any existing trails. He says, “So this was a novel therapy that had some good data, preliminary data about its anti-inflammatory effect and the vasodilator effect where it might improve patient’s oxygenation. So we were happy to use it. When we gave it to him initially, it took some doing to get because we have to clear it. So, when we gave it to the patient, it was three doses over three days. You’re hoping to see some kind of a side effect, which might suggest that the medication is working. He had none. Also, maybe an improvement in his oxygenation because if it’s a vasodilator, it might improve his oxygenation. It did not, which concerned me about its effect. But nonetheless, the patient did eventually recover.”
(Source: Daniel A. Gerardi, MD, FCCP Chief, Pulmonary, Critical Care, & Sleep Medicine, St. Francis Hospital, Hartford, CT
FOR MORE INFORMATION ON THIS REPORT, PLEASE CONTACT:
GREG PARASMO, DIR. OF COMMUNICATIONS, NEURO RX
(484) 254-6134
If this story or any other Ivanhoe story has impacted your life or prompted you or someone you know to seek or change treatments, please let us know by contacting Marjorie Bekaert Thomas at mthomas@ivanhoe.com