ORLANDO, Fla. (Ivanhoe Newswire) — Pulmonary embolism, or a potentially fatal blockage of blood vessels in the lungs, strikes as many as 600,000 Americans each year, many of whom had no symptoms. Now there is a new minimally-invasive treatment option that restores blood flow quickly.
Martha Hutchison, 73, cared for her late husband for five years then battled breast cancer, but it was an unexpected ticking time bomb that almost got the best of her.
“I remember turning this corner and heading to the bedroom and that’s the last thing I remembered. The next thing I knew I woke up and I was on the floor of the bedroom,” detailed Hutchison.
Paramedics rushed Hutchison to the hospital. She had no idea the pain in her leg she’d been ignoring was a clot. It was potentially blocking blood flow.
Rohit Bhatheja, M.D., FACC, an interventional cardiologist at Florida Hospital told Ivanhoe, “Unfortunately, she wasn’t a candidate for blood thinners.” (Read Full Interview)
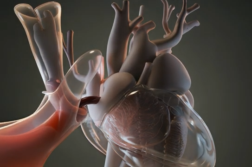
Instead, Dr. Bhatheja felt Hutchison would respond well to a new clot-removing device called the FlowTriever.
“FlowTriever is a mechanical mesh suction device,” said Dr. Bhatheja.
Doctors insert a catheter into the patient’s groin vein then move the device to the clot, catching the clot with the mesh, and then they retract the device.
Dr. Bhatheja explained, “We are not doing any major surgery. We are not cutting open any skin. We are doing everything through keyholes and punctures through the groin.”
Hutchison was released from the hospital after a few days, and has been slowly, but steadily on the mend.
“I can get up and do almost anything now,” said Hutchison.
Dr. Bhatheja said for some high-risk patients the FlowTriever procedure could be a better option than open cardiac surgery. He said patients can feel almost instant relief of the pressure on the heart and lungs.
Contributors to this news report include: Cyndy McGrath, Supervising and Field Producer; Milvionne Chery, Assistant Producer; Matt Goldschmidt, Editor; Tony D’Astoli, Videographer.
MEDICAL BREAKTHROUGHS – RESEARCH SUMMARY
TOPIC: FlowTriever Saves Lives
REPORT: MB #4168
BACKGROUND: Pulmonary embolism is a blockage in one of the pulmonary arteries in the lungs. It is caused by blood clots that travel to the lungs from the legs or other parts of the body. Since the clots block blood flow to the lungs, pulmonary embolism can be life-threatening, but prompt treatment greatly reduces the risk of death. Ten to 30 percent of people diagnosed with pulmonary embolism will die within a month. For 25 percent of cases, sudden death is the first symptom of pulmonary embolism.
(Source: http://www.mayoclinic.org/diseases-conditions/pulmonary-embolism/home/ovc-20234736)
SYMPTOMS AND RISK FACTORS: Symptoms for pulmonary embolism can vary greatly, but common symptoms include shortness of breath, chest pain and cough. Other symptoms include clammy of discolored skin, fever, excessive sweating, rapid or irregular heartbeat and lightheadedness or dizziness. Although anyone can develop blood clots and pulmonary embolism, there are certain risk factors that can increase your risk. A family history of blood clots or pulmonary embolism increases someone’s risk in addition to some medical conditions, such as heart disease and cancer. Surgery is also one of the leading causes of problem blood clots so medication to prevent clots may be given before and after major surgery to prevent this. Prolonged immobility that is often experienced during bed rest or long trips increases risk of blood clots, as well with smoking and pregnancy.
(Source: http://www.mayoclinic.org/diseases-conditions/pulmonary-embolism/symptoms-causes/dxc-20234744)
NEW TECHNOLOGY: Treatment options are limited for pulmonary embolism, especially for patients who are high-risk or cannot take blood thinners. However, a new device, called the FlowTriever, gives physicians a minimally-invasive way to restore blood flow in the lungs. FlowTriever is a catheter-based device inserted into the patient’s groin or leg vein. The physician guides the device to the clot, grabs hold of it with a medical mesh, and pulls the clot out of the body, providing almost instant relief of the pressure on the heart and lungs for patients. Most people are back home within two to three days of the procedure.
(Source: https://www.floridahospital.com/news/florida-hospital-patient-first-state-receive-revolutionary-treatment-pulmonary-embolism)
FOR MORE INFORMATION ON THIS REPORT, PLEASE CONTACT:
Amy Pavuk-Gentry
Corporate Communications Manager
Florida Hospital
407-303-1333
If this story or any other Ivanhoe story has impacted your life or prompted you or someone you know to seek or change treatments, please let us know by contacting Marjorie Bekaert Thomas at mthomas@ivanhoe.com