CLEVELAND, Ohio (Ivanhoe Newswire)— Heart devices, like pacemakers and defibrillators, are great at correcting heart rhythm problems. But they’re not without risk. Now, a new technology aims to make surgery for these devices safer and maybe stop heart infections.
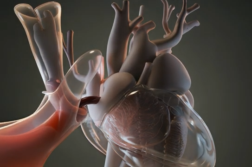
Every year, more than one million people worldwide receive an implantable heart device, like a pacemaker or defibrillator.
These complex gadgets can be lifesaving for some, but surgery to implant or upgrade them comes with a risk of infection.
Khaldoun Tarakji, MD, MPH of the Heart, Vascular, and Thoracic Institute at Cleveland Clinic stated, “Infection is not something you can treat with antibiotics alone, it’s the consequences of the infection that make us feel really concerned about it when it happens.”
A bad infection can lead to invasive surgeries, long hospital stays and even death. Typically, doctors give patients antibiotics before the procedure to prevent infection. But now, this antibiotic envelope can also be used during surgery. It’s made of an absorbable mesh that wraps around the device and stabilizes it. In a study of almost 7,000 patients, infection rates dropped by 40 percent when the antibiotic envelope was used.
“To prove that there’s an additional intervention, an additional therapy that you can provide as a preventive measure and was able to cut down the risk by 40 percent is something really remarkable,” Dr. Tarakji explained.
The envelope is coated with two antibiotics, minocycline and rifampin which are continually released over several days. It fully dissolves in the body after about nine weeks. A simple innovation that gives patients peace of mind when they need heart surgery.
Infections occur in about one to four percent of cardiac device implantations. The risk is particularly high following device replacements or other secondary procedures, like lead changes or upgrades.
Contributors to this news report include: Cyndy McGrath, Executive Producer; Julie Marks, Field Producer; Roque Correa, Editor.
To receive a free weekly e-mail on Medical Breakthroughs from Ivanhoe, sign up at: http://www.ivanhoe.com/ftk
MEDICAL BREAKTHROUGHS
RESEARCH SUMMARY
TOPIC: ANTIBIOTIC ENVELOPE STOPS HEART INFECTIONS
REPORT: MB #4768
BACKGROUND: An estimated 1.25 million permanent pacemakers are implanted globally each year and about 100,000 defibrillators are inserted in the U.S. each year. Arrhythmia is any disorder of your heart rate or rhythm and it means your heart beats too quickly, too slowly, or with an irregular pattern. If the arrhythmia is serious, you may need a cardiac pacemaker or an implantable cardioverter defibrillator (ICD). They are devices that are implanted in your chest or abdomen. A pacemaker helps control abnormal heart rhythms. It uses electrical pulses to prompt the heart to beat at a normal rate. It can speed up a slow heart rhythm, control a fast heart rhythm, and coordinate the chambers of the heart. An ICD monitors heart rhythm. If it senses a dangerous rhythm, it delivers shocks. This treatment is called defibrillation. An ICD can help control life-threatening arrhythmias, especially those that can cause sudden cardiac arrest. Most new ICDs can act as both a pacemaker and a defibrillator. Many also record the heart’s electrical patterns when there is an abnormal heartbeat which helps the doctor plan future treatment.
(Sources: https://medlineplus.gov/pacemakersandimplantabledefibrillators.html, https://www.mpo-mag.com/contents/view_online-exclusives/2020-02-21/examining-the-state-of-the-pacemaker-industry/, https://www.nytimes.com/2011/01/05/health/05device.html
HEART DEVICE INFECTIONS: Pacemaker and defibrillator pocket infections are a major complication after implantation. During implantation, there is a risk of device contamination with the patient’s own skin flora and it can be prevented by ideal surgical asepsis technique, pre and perioperative use of antibiotics. Most pacemaker infections occur after surgery. The infection rate is slightly higher for replacements. Most infections start in one of two places: either at the incision site or where the leads connect to the heart. Pacemaker-related infections are a special type of bacterial infection, called endocarditis. It’s the same life-threatening infection that affects the lining of your heart valves. These infections aren’t immediately lethal. Over time, however, the infection attacks your heart valves and can spread to your lungs and brain via the bloodstream. If it isn’t treated, it is sometimes fatal. The only treatment option is removal and replacement surgery. Your doctor will remove all the leads and any infected tissue. He or she will carefully remove any part of the device that has adhered to the heart tissue or veins to avoid tearing. The physician will implant a new device in a different location (such as in the opposite shoulder) and will prescribe a regimen of antibiotics.
(Sources: https://health.clevelandclinic.org/hidden-pacemaker-infection-making-sick/
NEW TECHNOLOGY: Khaldoun Tarakji, MD, Associate Section Head of Cardiac Electrophysiology at the Cleveland Clinic developed the antibiotic envelope for cardiac device implant surgery. The envelope is constructed of a multi-filament mesh that’s fully absorbable, and it has absorbable polymers that are mixed with 2 antibiotics: rifampin and minocycline. The envelope wraps the pacemaker or defibrillator before it’s implanted in the pocket. Then, the antibiotics get eluded in the pocket over a course of seven days. The envelope is fully absorbed within 9 weeks. It’s been shown to reduce the risk of major device-related infection by 40% within 1 year with no increase in complications.
(Source: https://www.mdmag.com/conference-coverage/acc-2019/khaldoun-tarakji-md-mph-wrapit-results
FOR MORE INFORMATION ON THIS REPORT, PLEASE CONTACT:
ANDREA PACETTI, PR
CLEVELAND CLINIC
(216) 316-3040
If this story or any other Ivanhoe story has impacted your life or prompted you or someone you know to seek or change treatments, please let us know by contacting Marjorie Bekaert Thomas at mthomas@ivanhoe.com