CLEVELAND, Ohio (Ivanhoe Newswire) — Right now, there are more than 378,000 clinical trials listed on the website clinicaltrials.gov. Millions of people look through the list each month to see if there is a new treatment, technology or procedure that can help them or their loved one. But how do you know which one is the right one for you? New technology is using genetic testing to make the perfect match.
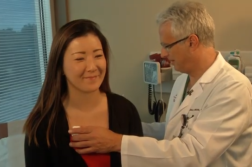
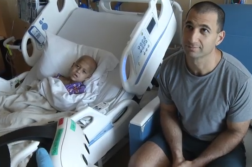
Andy Simon is a proud cancer survivor, ribbon wearing man! Andy was diagnosed with glioblastoma multiforme, one of the deadliest forms of brain cancer.
“He doesn’t have long. That was my first thought,” explained Amy Simon, Andy’s wife.
But after surgery and taking part in a novel clinical trial, five years out, there’s no re-occurrence of Andy’s cancer.
“There was no doubt in my mind that the clinical trial helped him,” continued Amy.
But there are more than 1,600 clinical trials happening right now for glioblastoma patients.
“It’s not so much about rushing the treatment but rushing to the right treatment,” shared Joel Saltzman, MD, a medical oncologist at University Hospitals Seidman Cancer Center in Cleveland, Ohio.
New technology called genomoncology precision decision works like a match maker, taking the genetic sequencing of the patient and running it through a database.
“We can look at numerous genes that predict what might respond to therapies. Rather than telling them about the standard treatment, I actually know walking in the door that, hey, we have this great study,” said Dr. Saltzman.
Andy and his wife believe his clinical trial saved his life.
“I believe in my heart that, all of that, he went through is the reason that he’s here today,” Amy stated.
To celebrate his last treatment Andy put on a cape to proudly show his superman status.
“My famous line is now set to kick its ass. And we’ve been doing it since, as a team,” exclaimed Andy.
And they’ll continue to kick cancer together.
Experts believe the genomoncology precision decision will also help to increase the clinical trial success rates by matching the right patients with the right therapies. The match maker works on all types of cancers.
Contributors to this news report include: Marsha Lewis, Producer; Roque Correa, Editor; and Kirk Manson, Videographer.
CLINICAL TRIAL MATCH MAKER REPORT #2867
BACKGROUND: Clinical trials are research studies performed in people that are aimed at evaluating a medical, surgical, or behavioral intervention. They are the main way that researchers find out if a new treatment, like a new drug, or diet, or medical device is safe and effective in people. A clinical trial is often used to learn if a new treatment is more effective and/or has less harmful side effects than the standard treatment. Before the FDA approves a clinical trial to begin, scientists perform lab tests and studies in animals to test a potential therapy’s safety and efficacy.
(Source: https://www.nia.nih.gov/health/what-are-clinical-trials-and-studies)
FINDING A CLINICAL TRIAL: In the past, clinical trials were done only at major medical centers. So, patients had to travel a long way and be treated by doctors they didn’t know. There are still some Phase I and Phase II studies where this is still the case. Patients have more options today including staying closer to home or even staying with their own doctors during a study. There are groups that provide ways to search for clinical trials on their websites, and many of these groups also have people who can help you with the search. Search results often include a description of each study, eligibility factors that people must meet to go into the trial, and a contact person. A new technological platform through GenomOncology’s Precision Decision and GO Connect solutions, streamlines the clinical trial process in order to help both patients and doctors. It provides the capability to identify groups of patients within their institution eligible for clinical trials and novel therapies.
(Source: https://www.cancer.org/treatment/treatments-and-side-effects/clinical-trials/what-you-need-to-know/picking-a-clinical-trial.html and https://news.uhhospitals.org/news-releases/new-technology-matching-patients-genomic-testing-with-available-clinical-trials-nationwide-arrives-at-university-hospitals.htm)
ADVANCES IN DNA SEQUENCING: Nanopore technology shows promise for making it possible to develop small, portable, inexpensive devices that can sequence DNA in real time. Researchers at The University of Texas at Dallas, have moved closer to developing a nanopore sequencing platform that can detect the presence of nucleobases, the building blocks of DNA and RNA. “By enabling us to detect the presence of nucleobases, our platform can help improve the sensitivity of nanopore sequencing,” said Dr. Moon Kim, professor of materials science and engineering and the Louis Beecherl Jr. Distinguished Professor in the Erik Jonsson School of Engineering and Computer Science. One of the challenges has been the difficulty in controlling the speed of the DNA strand as it moves through the nanopore. The team’s research focused on addressing that by fabricating an atomically thin solid-state membrane coated with titanium dioxide, water and an ionic liquid to slow the speed of the molecules through the membrane. The water was added to the liquid solution to amplify the electrical signals, making them easier to read. “The ultimate goal is to have a hand-held DNA sequencing device that is fast, accurate and can be used anywhere,” Kim said.
(Source: https://www.sciencedaily.com/releases/2021/04/210429104936.htm)
* For More Information, Contact:
Jeannine Denholm, Sr. Media Relations Strategist Joel Saltzman, MD
jeannine.denholm@uhhospitals.org UH Seidman Cancer Center
(216) 844-2555 (440) 975-7589
Free weekly e-mail on Medical Breakthroughs from Ivanhoe. To sign up: http://www.ivanhoe.com/ftk